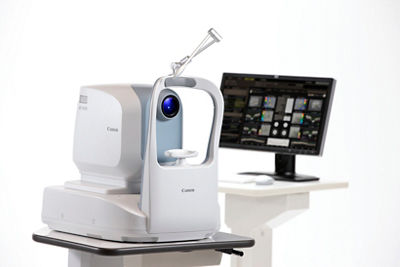
Canon Xephilio OCT-A1 Optical Coherence Tomography Device Receives FDA 510(k) Clearance
Commercially available, bringing an intuitive, user-friendly design to the market
MELVILLE, NY, September 17, 2019 – Expanding the company’s lineup of eye-care product offerings, Canon U.S.A., Inc., a leader in digital imaging solutions, is pleased to announce the new intuitive, user-friendly Xephilio OCT-A1 Optical Coherence Tomography device from Canon Inc. This device features automated acquisition functionality, which makes it easy to use, allowing healthcare professionals to obtain high-quality reproducible scans and images. The Xephilio OCT-A1 Optical Coherence Tomography device has received FDA 510(k) clearance for sale in the United States of America1.
The Xephilio OCT-A1 Optical Coherence Tomography device, together with the required RX Capture software, computer and LCD monitor (collectively, the “Xephilio OCT-A1 System”), is an optical coherence tomography system indicated for the in-vivo imaging and measurement of the retina, retinal nerve fiber layer, and optic disc. In addition, it is a tool to aid in the diagnosis and management of retinal diseases by a clinician. The Xephilio OCT-A1 System has a scanning speed of 70,000 A-scans per second with optical depth resolution of approximately three microns. With its integrated scanning laser ophthalmoscopic real-time retinal tracking technology, the Xephilio OCT A-1 System automatically retains the scan position and scan protocol for each patient from one exam to the next, eliminating the need for manual adjustment by the operator. A mouse-controlled, three-click operation is used to engage the automated alignment, tracking, and acquisition of high-quality images and scans. To help create a positive, effective user experience, the positioning of the components of the Xephilio OCT-A1 System can allow for the operator and patient to sit side-by-side.
“Designed to meet the needs of customers, the Xephilio OCT-A1 System is a user-friendly solution designed to help healthcare professionals capture high-resolution images and scans with ease and efficiency,” says Tsuneo Imai, vice president and general manager, Healthcare Solutions, Business Information Communications Group, Canon U.S.A., Inc. “Canon U.S.A. is proud to make the Xephilio OCT-A1 System available to its customers.”
The Xephilio OCT-A1 System is versatile and can be used by healthcare professionals in a variety of settings, including ophthalmology private practices, optometric schools, telehealth, and telemedicine primary care providers2. In addition, the Xephilio OCT-A1 System can also help healthcare professionals identify the disease state and monitor disease conditions before and after treatments in ophthalmology private practices, retinal clinics, teaching hospitals, and institutions. For optometry practices and optometric schools, this System can aid optometry students in recognizing diseases and conditions in primary care, while also showing them how to detect conditions prior to and after referral to ophthalmology practitioners.
“The Xephilio OCT-A1 System with its easy-to-use functions (three mouse clicks to capture any OCT image through a minimum of 3.0 mm pupil) is a great system for medical and optometry students as it delivers high-quality imagery with approximately three micron optical depth resolution and 70,000 scans,” says Ben Szirth, Ph.D., Director, Tele-Ocular Program, Institute of Ophthalmology and Visual Science, New Jersey Medical School3. “It is this type of system that may assist students preparing to enter the competitive field of eye care.”
The Xephilio OCT-A1 System will be showcased in the Canon Booth #MS9048 at Vision Expo West, taking place from September 19-21, 2019 at the Sands Expo & Convention Center in Las Vegas. In addition to the Xephilio OCT-A1 System, this booth will also exhibit the CR-2 PLUS AF Digital Non-Mydriatic Retinal Camera.
For more information, please call (800) 970-7227, or visit usa.canon.com/eye-care.
About Canon U.S.A., Inc.
Canon U.S.A., Inc., is a leading provider of consumer, business-to-business, and industrial digital imaging solutions to the United States and to Latin America and the Caribbean markets. With approximately $36 billion in global revenue, its parent company, Canon Inc. (NYSE:CAJ), ranks third overall in U.S. patents granted in 2018† and was named one of Fortune Magazine's World's Most Admired Companies in 2019. Canon U.S.A. is dedicated to its Kyosei philosophy of social and environmental responsibility. To keep apprised of the latest news from Canon U.S.A., sign up for the Company's RSS news feed by visiting www.usa.canon.com/rss and follow us on Twitter @CanonUSA.
# # #
† Based on weekly patent counts issued by United States Patent and Trademark Office.
1 FDA clearance does not in any way denote FDA approval of this device.
2 Requires both senders and receivers to have internet access and the recipient to have an operating system that can open jpeg or pdf files.
3 Ben Szirth, Ph.D., is a paid consultant who provides advisory and consulting services to Canon U.S.A. on a regular basis.